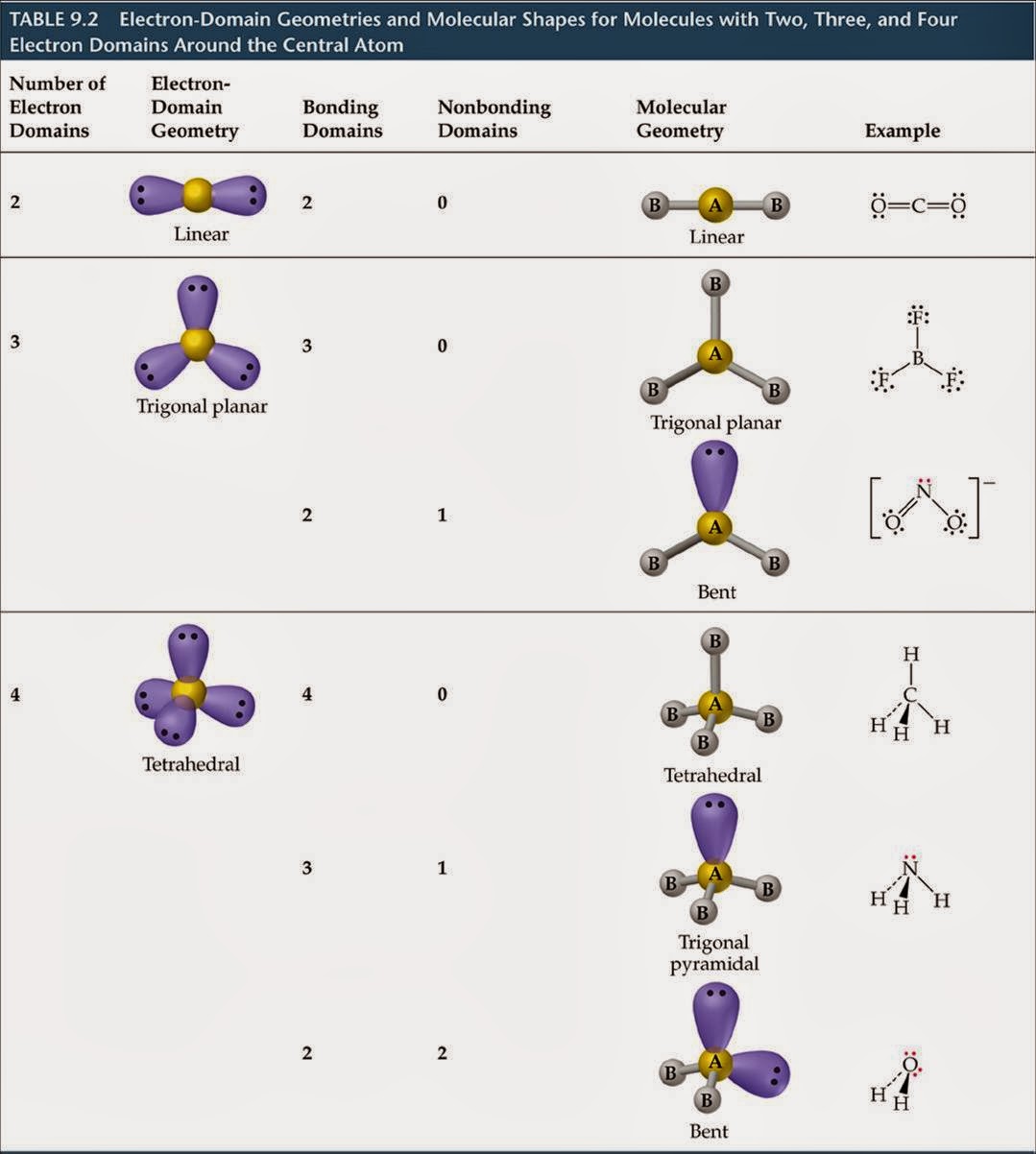
" Chemical Bonding", Oxford University Press, 2nd edition, 2016, ISBN: 978-0-19-870095-1.Ĭhemputer, a set of simple calculators for chemistry: Ĭopyright 1993-2023 Prof Mark J. The VSEPR calculation used here is described in detail within the Oxford Primer: Mark J. Compare bond angle predictions from the VSEPR-based model to real molecules. Name molecule and electron geometries for molecules with up to six electron groups surrounding a central atom. Recognize the difference between electron and molecular geometry. 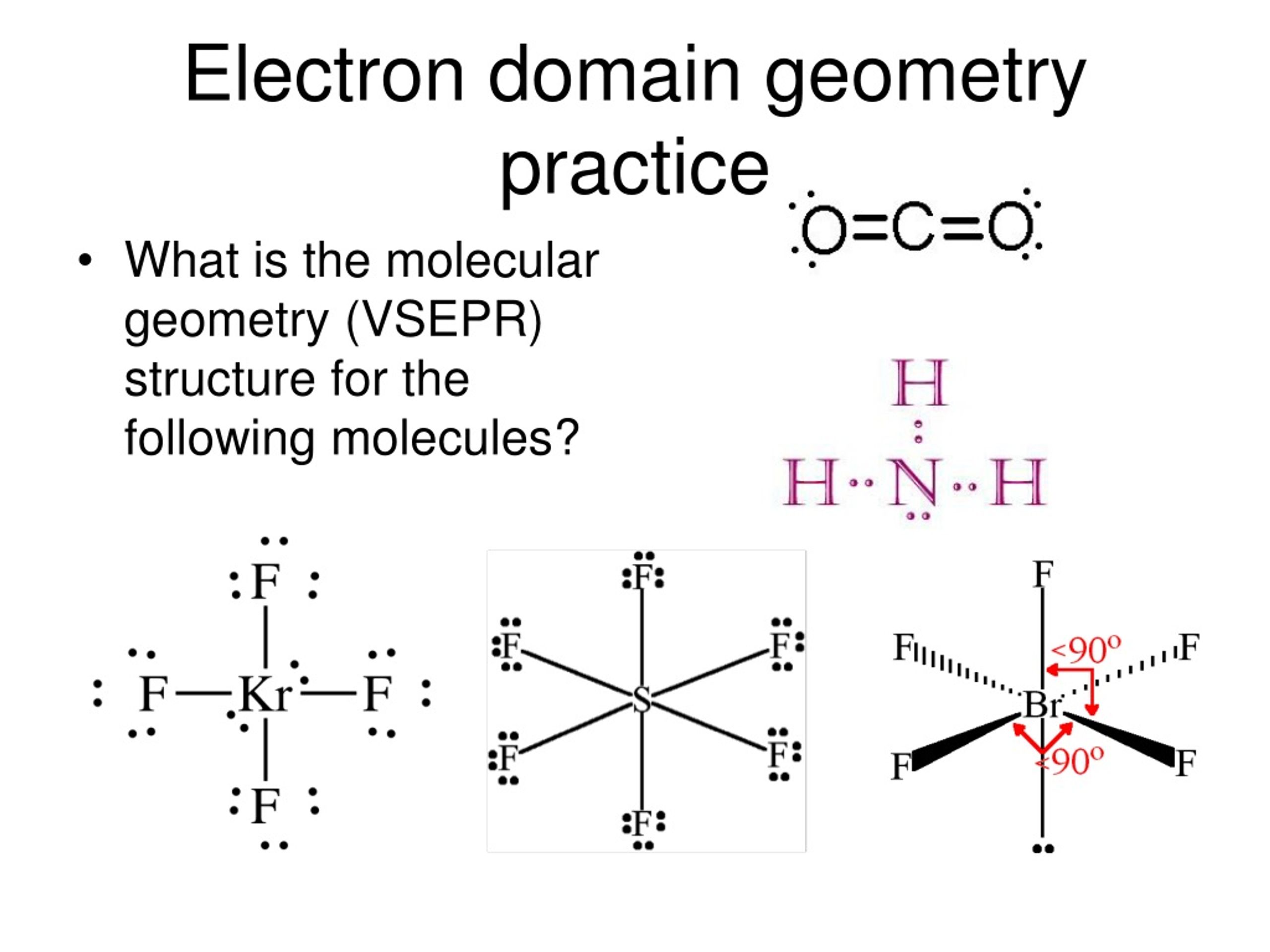
Radicals are not handled well at present. Recognize that molecule geometry is due to repulsions between electron groups.It describes the geometry of only the sigma bonds, and unfortunately you have to memorise the names of these (both electron domain and molecular) geometries. Two pairs associated with octahedral σ-frameworks are genrerally trans. calpers retirement calculator table 2 at 55 0. The molecular geometry is trigonal pyramidal (cause it looks like a pyramid with three legs lmao).Lone pairs associated with trigonal bipyramidal σ-frameworks are generally axial.If you build it with two oxo O ⊖ groups and one =O group you will also get the correct answer but do make sure to include the charge on the compound of -2. If you build it with three =O groups and a charge on the compound of -2 you will get the right answer. Suppose you wanted to build carbonate, 2–. The Chemputer will then predict the geometry for your chosen central atom. You must define the atom of interest (the central atom), the overall charge, and at least one attached group.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
